Projects
We use a combination of cellular, molecular and genetic tools to study neurodevelopment using in vitro models.
Our Projects
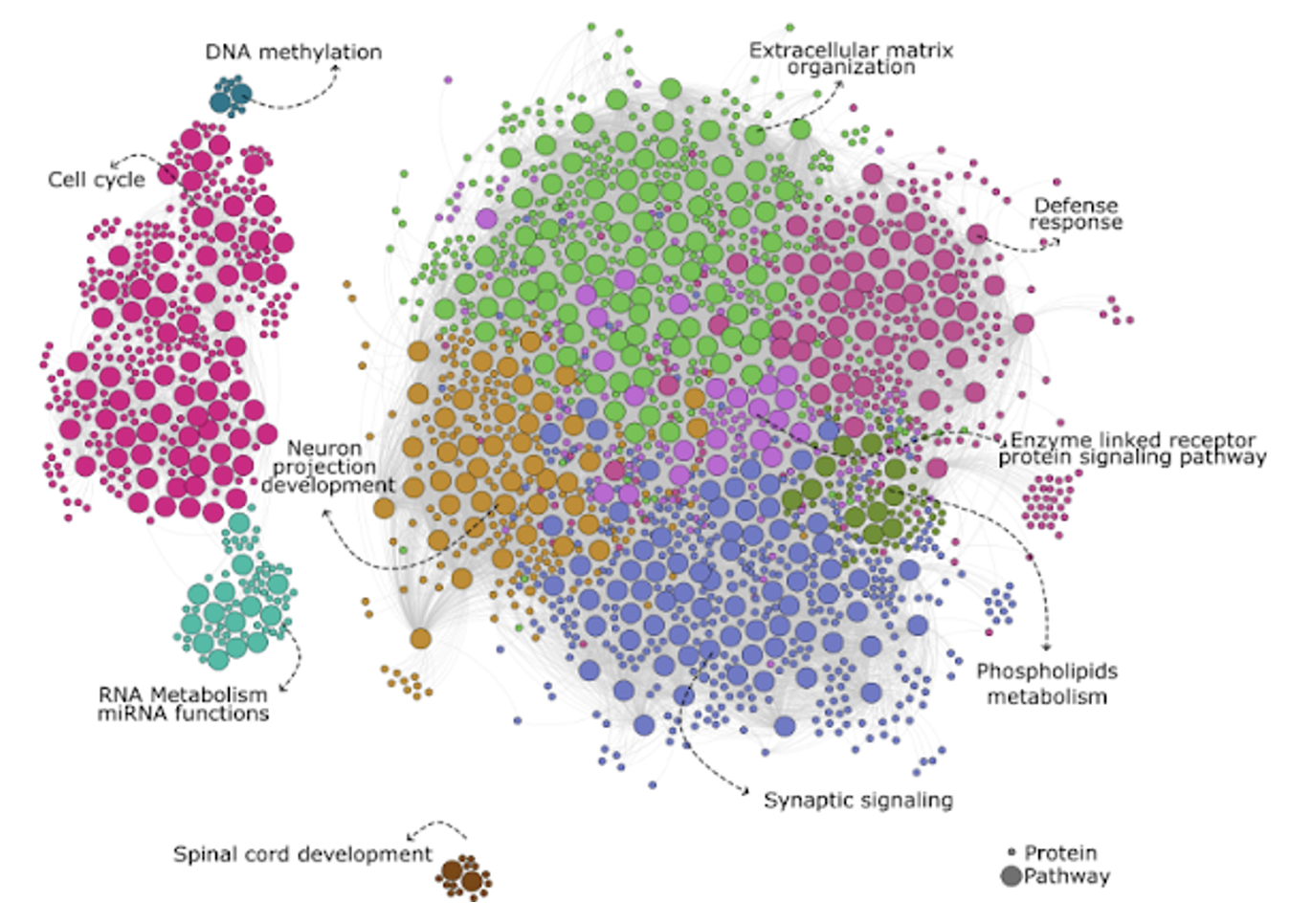
The Autism Susceptibility Candidate 2 (AUTS2) gene has emerged as a critical regulator of neurodevelopment and is strongly associated with a wide range of neurological and neuropsychiatric disorders, including autism spectrum disorder (ASD). Individuals with AUTS2 mutations often exhibit microcephaly, craniofacial abnormalities, and repetitive behaviors. In addition, genetic variations within the AUTS2 locus have been linked to diverse conditions such as epilepsy, schizophrenia, attention-deficit/hyperactivity disorder (ADHD), dyslexia, depression, and substance use disorders. To investigate the role of AUTS2 in human brain development, we utilize human stem cell-derived models in combination with CRISPR/Cas9 gene editing. We employ next-generation sequencing to define AUTS2-mediated gene regulatory networks and use live-cell imaging assays to examine the effects of AUTS2 loss on brain organoid development, neuronal differentiation, morphogenesis, and maturation. Together, these approaches aim to uncover the molecular and cellular mechanisms by which AUTS2 contributes to human neurodevelopment and disease.
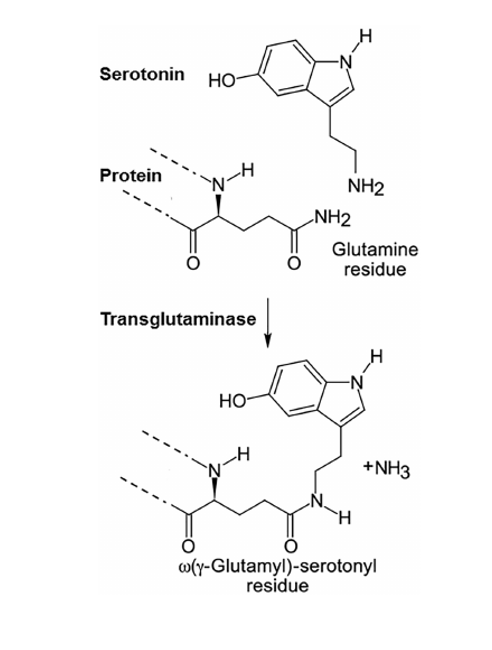
Prenatal selective serotonin reuptake inhibitor (SSRI) exposure has been linked to increased risk for autism spectrum disorder (ASD), but the effects of SSRI exposure on histone serotonylation have not yet been characterized in ASD. Using an antibody for the dual modification histone 3 lysine 4 trimethylation adjacent to glutamine 5 serotonylation (H3K4me3Q5ser) we are studying the effects of histone serotonylation on gene expression in SSRI-treated ASD and controls. This study characterizes the relationship between genetic background, SSRI exposure, histone serotonylation, and gene expression in induced pluripotent stem cells and neural progenitor cells. These results are valuable for assessing histone serotonylation as a potential target for ASD prevention, diagnostics, and therapeutics.
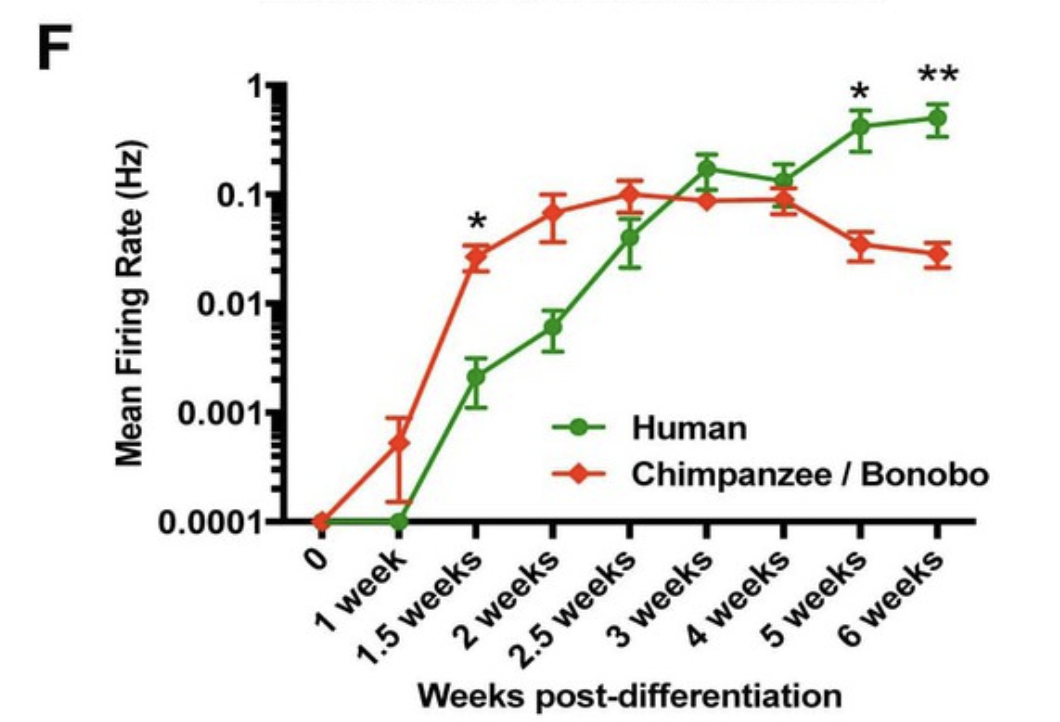
Human neurons are characterized by a slower developmental trajectory than non-human primate neurons. We are interested in studying the regulatory mechanisms that set development rate across species.
Polystyrene micro- and nano-plastics (PS-MNPs) are found not only in individuals with occupational exposure but also widely in the general population, including in drinking water. They can enter the human body through ingestion, inhalation, and skin contact, and have been detected in human blood, urine, breastmilk, and placental tissue. Studies suggest that MNPs may reach the fetal brain, posing risks to neural development, particularly during pregnancy. Evidence links MNP exposure to reduced fetal birth weight, neuroinflammation, oxidative stress, and disrupted neurogenesis. We utilize neural organoids to model how PS-MNPs affect fetal brain development and neuronal function, aiming to better understand the timing and mechanisms of their impact on early human neurodevelopment.
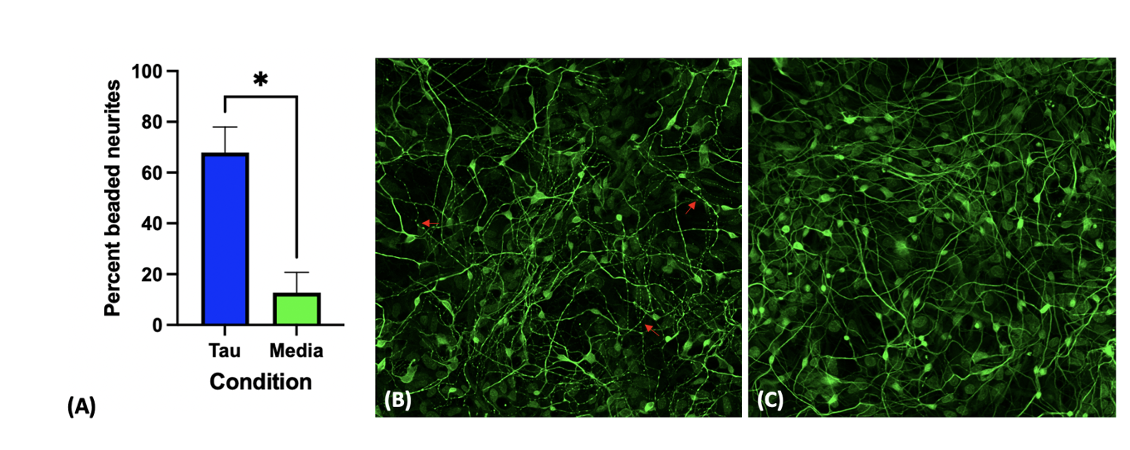
Primates—and humans in particular—rely heavily on memory and cognitive flexibility to navigate complex physical and social environments. This cognitive demand is thought to have played a significant role in the evolutionary expansion of the human brain. However, the same neural adaptations that support advanced cognition may also have rendered the human brain more vulnerable to age-related neurodegenerative diseases. Alzheimer’s disease (AD), the most common form of age-dependent dementia, exemplifies this paradox. Despite extensive research, the underlying causes and mechanisms of AD remain incompletely understood. Moreover, while nonhuman primates (NHPs) share many neurological features with humans, studies in NHPs have yielded inconclusive evidence regarding the full expression of AD pathology. To explore the evolutionary underpinnings of neurodegeneration, we use induced pluripotent stem cell (iPSC)-derived neurons from both humans and nonhuman primates. This comparative approach enables us to investigate species-specific molecular and cellular mechanisms associated with brain aging and susceptibility to neurodegeneration. Ultimately, this research aims to illuminate the evolutionary differences that may underlie the unique vulnerability of the human brain to diseases like Alzheimer’s.